Researchers in China have identified a groundbreaking method to convert greenhouse gases, specifically carbon dioxide (CO2) and methane (CH4), into valuable chemicals without the need for costly catalysts. This process utilizes high-energy photons generated by a specialized ultraviolet light source, marking a significant advancement in climate change mitigation.
The study, published on December 14, 2025, in the journal Nature Photonics, reveals that photons with a wavelength of 185 nm effectively break the strong chemical bonds in both CO2 and CH4. The researchers achieved this using a 28-W ultraviolet light source, allowing them to transform these gases into useful compounds such as carbon monoxide and ethane under ambient conditions.
Climate Challenges and Innovative Solutions
Carbon dioxide and methane are responsible for approximately 84% of the global rise in temperatures, positioning them as major contributors to climate change. CO2 is also the leading factor in ocean acidification, which poses a significant threat to marine ecosystems. In response to these pressing environmental challenges, scientists are exploring methods to not only reduce emissions but also to repurpose existing greenhouse gases into beneficial products.
Traditionally, converting CO2 and CH4 into different molecules has been a complex task. Their chemically stable nature, characterized by strong bonds, renders them relatively inert. Conventional approaches often rely on expensive metal catalysts and require extreme temperatures exceeding 700 °C and high pressures, resulting in energy-intensive and costly processes.
Innovative Light-Driven Conversion Process
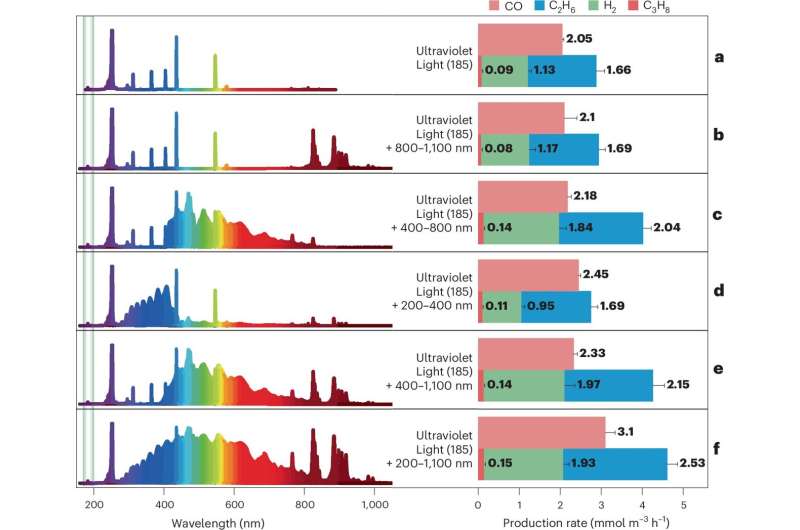
The research team constructed a quartz reactor chamber filled with a mixture of 99.9% pure CO2 and CH4. By exposing this mixture to various light sources under controlled conditions—specifically, a temperature of 25°C—they found that high-energy photons at 185 nm could initiate the conversion process.
Results from gas analysis indicated the production of carbon monoxide, hydrogen, and ethane, with rates of 3.1 mmol m-3 h-1, 1.93 mmol m-3 h-1, and 2.53 mmol m-3 h-1 respectively. The research showed that enhancing the mixture with water and removing atmospheric oxygen improved yields.
In an impressive simulation of outer-space conditions by flushing the reactor with argon gas, the team achieved a total gas conversion of 1.51% within 24 hours. While the current yield is modest, the researchers emphasize that their findings demonstrate a promising new approach to converting two prevalent greenhouse gases into valuable products using only light and standard environmental conditions.
This innovative method represents a crucial step toward building a circular economy, wherein waste materials can be transformed into useful resources, ultimately contributing to sustainability efforts. The implications of this research could be far-reaching as scientists continue to seek effective solutions to combat climate change.
The article underscores the collaborative effort of the research team, led by Jianxin Zhai, with content reviewed by Robert Egan and edited by Gaby Clark. This work exemplifies the ongoing commitment to advancing scientific knowledge and addressing pressing global issues through innovative research.